AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Density of water in kgm34/11/2024 Thanks to the density conversion calculator, we can see that 1 g/cm 3 is equal to 1,000 kg/m 3 but also 8,345 lb/gal or 0.578 oz/cu in. Grams per cubic centimeter is one of the standard units used around the world to express density. This form of the equation is handy for kitchen recipes conversion - check out our great grams to cups calculator, and recipes from different countries won't be a problem anymore. Given the density of a product and its weight in grams, you can easily find its volume. 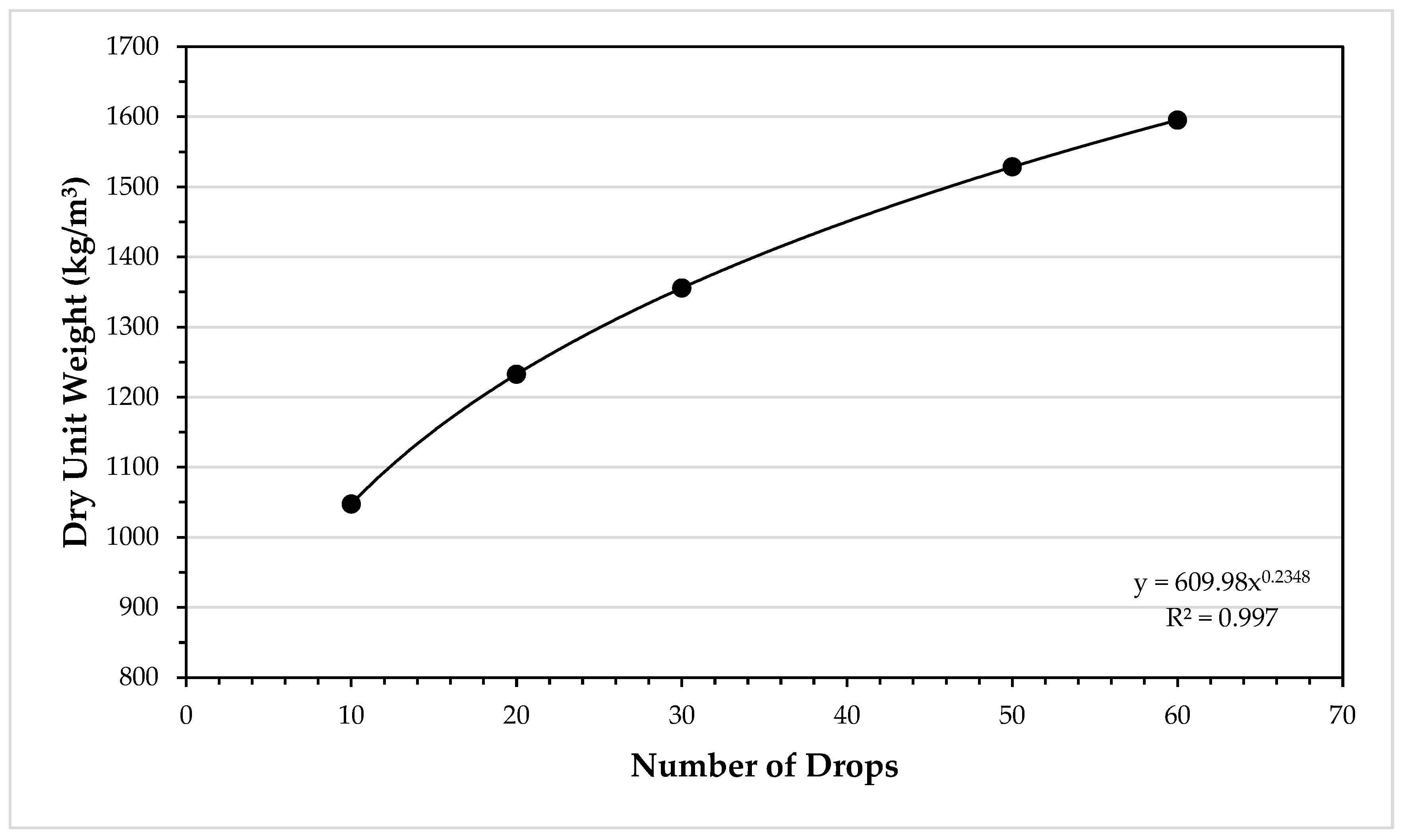
The density equation may also be useful in everyday life. A similar tool also available on our platform is the density calculator. The density of water in lb/gal, or, if, using metric system, the density of water in grams per cubic centimeter can also be calculated using this tool. In general the value is relatively large for solids and fluids while it tends to be smaller for gases. Density can be measured for any substance and the results will vary depending on its temperature, pressure, buoyancy, purity and packaging, to name a few factors. It's usually expressed by the Greek letter ρ and expressed in various density units. Density itself is defined as how much mass a substance has in a specific volume. Table 14.1 - Densities of Some Common Substances SolidsĪs you can see by examining Table 14.Density conversion tool enables you to quickly calculate the weight to volume ratio of an object in any of the available units. Plasma will not be discussed in depth in this chapter because plasma has very different properties from the three other common phases of matter, discussed in this chapter, due to the strong electrical forces between the charges. At high temperatures, molecules may disassociate into atoms, and atoms disassociate into electrons (with negative charges) and protons (with positive charges), forming a plasma. There exists one other phase of matter, plasma, which exists at very high temperatures. In this chapter, we generally refer to both gases and liquids simply as fluids, making a distinction between them only when they behave differently. When placed in an open container, gases, unlike liquids, will escape. This makes gases relatively easy to compress and allows them to flow (which makes them fluids). 
In contrast, atoms in gases are separated by large distances, and the forces between atoms in a gas are therefore very weak, except when the atoms collide with one another. Because the atoms are closely packed, liquids, like solids, resist compression an extremely large force is necessary to change the volume of a liquid. When a liquid is placed in a container with no lid, it remains in the container. That is, liquids flow (so they are a type of fluid), with the molecules held together by mutual attraction. This occurs because the atoms or molecules in a liquid are free to slide about and change neighbors. Liquids deform easily when stressed and do not spring back to their original shape once a force is removed. A gas must be held in a closed container to prevent it from expanding freely and escaping. 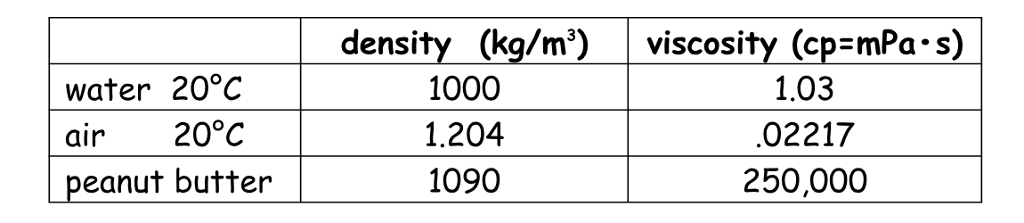
(c) Atoms in a gas move about freely and are separated by large distances. Forces between the atoms strongly resist attempts to compress the atoms. (b) Atoms in a liquid are also in close contact but can slide over one another. \): (a) Atoms in a solid are always in close contact with neighboring atoms, held in place by forces represented here by springs.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
